Studying synapses in specific neural circuits
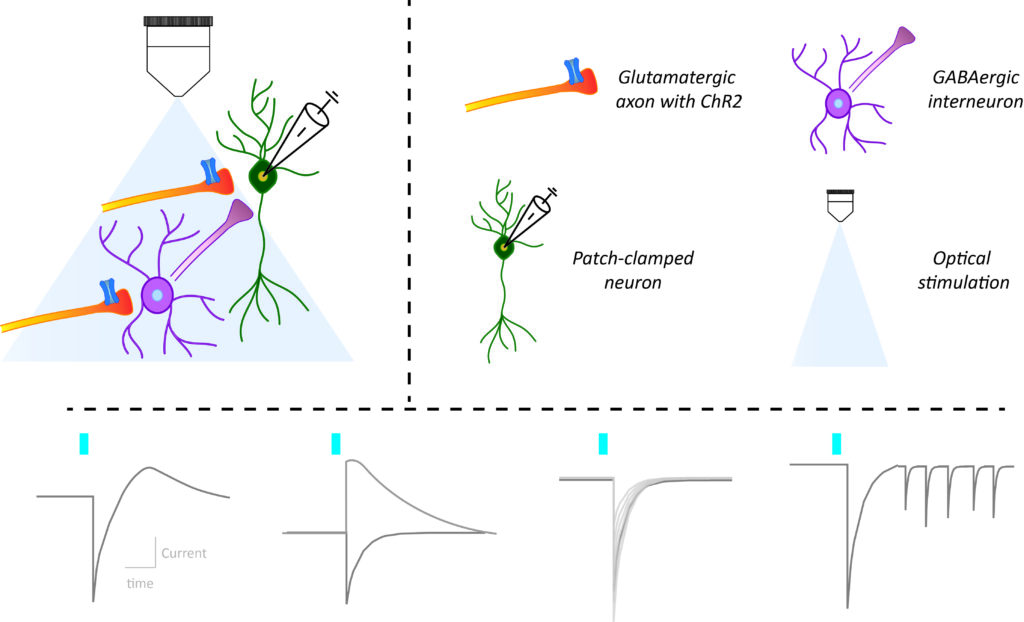
In recent years the combination of brain slice electrophysiological recordings of neurons in response to optogenetic stimulation of their input, has become an indispensable staple to probe the function of neural circuits. The large amounts of distinct metrics that one can obtain this way are both informative but also daunting to the newcomer. In a recent review paper, Frank Meye’s team summarized the methodological state-of-the-art for this approach. The paper featured several team members including co-first authors Laura Supiot and Louisa Linders. In particular, the authors reviewed the rationale behind the different metrics used to study synaptic connectivity and changes in synaptic strength. They compiled a guide for the implementation of these methodologies in practice and discussed future directions to decipher neural circuits.
“We aimed to provide the rationale for distinct electrophysiological synaptic metrics, as well as practically explain how to obtain them.” Laura Supiot
Also see the tweet by Frank Meye. Congratulations to the whole team! 🎉
Renata Vieira de Sá finalises her PhD on ALS


Another successful PhD on ALS! On Thursday 20th of October Renata Vieira de Sa defended her PhD thesis “Repeat expansions in Amyotrophic lateral sclerosis (ALS). Lessons from patient-derived models” at the Academiegebouw of Utrecht University. In her PhD thesis, Renata studied the contribution of C9orf72 and Ataxin2 repeat expansion to ALS using different patient-derived models including brain organoids and 2D motor neurons. She was supervised by promotors Jeroen Pasterkamp and Leonard van den Berg. This PhD on ALS further solidifies UMC Utrecht Brain Center as a leader in the field. Her work on human organoid models was instrumental in developing the cellular diversity. Renata continues her career as a post doctoral researcher at uniQure B.V. and the OrganoVIR labs in the AMC where she will continue developing advanced human models.
“It was a pleasure to defend my thesis after working on this topic for so long! It was a great way to conclude my time at the brain Center ” Renata
Once again, congratulations!
PhD defense of Christiaan Huffels

On Tuesday October 18, Christiaan Huffels successfully defended his PhD thesis, titled “Tracing the signals: Unravelling neuron-glia interactions in Alzheimer’s disease”, at the Academiegebouw of Utrecht University. In his thesis, Christiaan examined the role of astrocytes and microglia in the early development of Alzheimer’s disease using a multidisciplinary approach, combining the use of slice electrophysiology, calcium imaging, immunohistochemistry, behavioral analyses, and pharmacological interventions. He was supervised by promotor Prof. dr. Elly Hol and co-promotor Dr. Jinte Middeldorp. For his next career step, he will continue his work as a postdoc in the lab of Prof. dr. Elly Hol at the University Medical Center Utrecht Brain Center, where he will focus on developing functional assays on human cell culture systems.
“I enjoyed working on this interesting topic for 4.5 years and I am grateful to have ended it in such a great way! It was a great day and I’m happy to continue working in the Hol lab to further develop my skills as a researcher” Christiaan Huffels

The work for his thesis was performed at the Translational Neuroscience department. His output include articles titled “Aß Pathology and Neuron-Glia Interactions: A Synaptocentric View” published at the Neurochemical Research and “Systemic Injection of Aged Blood Plasma in Adult C57BL/6 Mice Induces Neurophysiological Impairments in the Hippocampal CA1” published at Journal of Alzheimer’s Disease. We are looking forward to his contribution to the scientific research.
GliaNed meeting 2022
Big success with the annual GliaNed meeting! The main goals of this informal meeting are to stimulate interaction between glia researchers in The Netherlands and to exchange knowledge. Elly Hol (translational neuroscience, UMCU) and Wia Baron (UMCG) put together a great program and were thrilled to have Prof. Inge Huitinga to open the show with her keynote lecture on ‘The role of microglia in MS and major depression’.
The meeting got additional contribution from our department. Carla Gomes da Silva presented her work on how oligodendrocyte precursors regulate cortical networks during mammalian development. Werner Dykstra explained how he studies the role of the astrocyte cytoskeletion in brain organoids. Several other researchers throughout the Netherlands have presented their latest research.

“It is very impressive how glia is involved practically in every brain process and disease you can imagine. GliaNed is providing an essential platform to bring researchers of this rapidly growing field together” Onur Basak
We hope to see the community at the next GliaNed meeting in Groningen.
Inauguration of Elly Hol as a KNAW member

 We proudly celebrate the inauguration of prof. Elly Hol of our Translational Neuroscience department as a Royal Netherlands Academy of Arts and Sciences (KNAW) member 🎊! She is one of the 22 new members who were at the historic Trippenhuis this Monday. This is a great honor given to a select group of scientists in the Netherlands
We proudly celebrate the inauguration of prof. Elly Hol of our Translational Neuroscience department as a Royal Netherlands Academy of Arts and Sciences (KNAW) member 🎊! She is one of the 22 new members who were at the historic Trippenhuis this Monday. This is a great honor given to a select group of scientists in the Netherlands
Elly Hol external link is Professor of Gliabiology of Brain Diseases and one of the heads of the Translational Neuroscience Department at the UMC Utrecht Brain Centre. Tained as a medical biologist with a specialisation in molecular neurobiology, she obtained her PhD in Utrecht. Next, she worked at the Max-Planck-Institute for Psychiatry in Martinsried with a Max-Planck Fellowship to. From 2013 onwards, Elly has worked as a principal investigator. In 2015, whe was appointed Professor of Gliabiology of Brain Diseases at the Utrecht University. She is a member of the Academia Europaea, chair of the scientific advisory board of Parkinson Netherlands, chair of GliaNed, programme leader of the Neuroscience master’s and PhD programme at Utrecht University Graduate School of Life Sciences, and education manager at the Brain Division of UMC Utrecht. Her inauguration was the results of years of contribution to the establishment of glia biology.
“Elly Hol belongs to the still small but growing group of neuroscientists who focus on the biology of glia. These are non-neuronal cells that play a crucial role in keeping our brain healthy. They act as stem cells and immune system, but can also derail. Hol has published a number of highly cited articles, in which she uncovers a correlation between certain proteins in the human brain and glial cells that play a role in the development of brain tumors and diseases such as Alzheimer’s and epilepsy. Hol is an important source of inspiration for young researchers, including through the national Glia network she leads and through personal mentorships for young female researchers.” KNAW
You can find more information on her research at our website and her lab page. More information on the inauguration is available on the Utrecht University and KNAW websites.